Cellusion Receives U.S. FDA Orphan Drug Designation for CLS001
Cellusion Inc. (CEO: Shin Hatou, M.D. Ph.D., “Cellusion”), a deep tech start-up developing a novel cell therapy, announced that the U.S. Food and Drug Administration (“FDA”) has granted Orphan Drug Designation (“ODD”) to our regenerative medicine product “iPS cell-derived corneal endothelial cell substitute” (“CLS001”) for the treatment of bullous keratopathy.
FDA grants ODD if the drug meets specific criteria, the treatment, diagnosis, or prevention of rare diseases that affect less than 200,000 people in the United States. The ODD qualifies sponsors for incentives, including tax credits for qualified clinical (in humans) testing, waiver of the prescription drug user fee and potential 7 years of market exclusivity after FDA approval.
Cellusion has developed CLS001 for bullous keratopathy, which accounts for more than half of all corneal transplants and is currently preparing for corporate clinical trials in Japan and global clinical trials. With this designation, we will further accelerate the global development of CLS001 and aim to contribute to patients around the world who are waiting for the treatment.
The ODD information for CLS001 is as follows:
FDA (Search Orphan Drug Designations and Approvals)
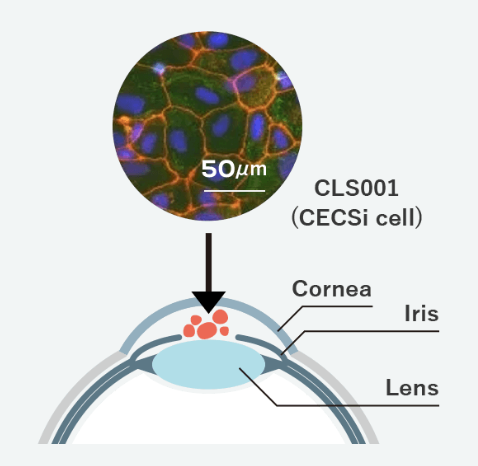
[About CLS001]
CLS001 is alternative cell to corneal endothelial cells produced by efficiently differentiating iPS cells, which can be differentiated into various types of cells, based on Cellusion’s proprietary technique. Cellusion is promoting the development of CLS001 as a therapeutic regenerative medicine product for bullous keratopathy, which accounts for more than half of the corneal transplant cases, by combining “CECSi Cells* made from iPS cells with excellent proliferative properties” and “a simple injection cell delivery procedure without needs of human expertise.” Cellusion is also advancing preparations for company-sponsored clinical trials in Japan, as well as preparations for global clinical trials.
*CECSi Cells: Corneal Endothelial Cell Substitute from iPS Cells
[About Bullous Keratopathy]
Bullous keratopathy is a rare disease that occurs when corneal endothelial cells, which control the amount of fluid at the inner-most part of the cornea, decrease due to eye surgery of cataract or genetic factors, resulting in problems with their function, whereby the cornea becomes swollen and causes visual impairment due to opacity. This disease is progressive and may lead to blindness if left untreated. Today, this disease is mainly treated by transplanting normally functioning corneal endothelial cells via corneal transplant. Nevertheless, as with other organ transplants, corneal transplants are also facing a serious problem of donor shortage. In Japan alone, it is estimated that there are about 10,000 patients, and in the global survey conducted in 2013 it was reported that approximately 13 million patients are waiting for corneal transplants.
[About Cellusion]
Cellusion is a regenerative medicine start-up originating from the Department of Ophthalmology, Keio University School of Medicine, with “Regenerating Human Potential” as its mission and “More Freedom and More Smiles to the World” as its vision. Based on patented technology such as its proprietary technique of efficiently producing corneal endothelial substitute cell from iPS cells, Cellusion is promoting R&D aimed at meeting medical needs from the perspective of “patient centricity” by leveraging cutting-edge regenerative medicine technology, including the lead program CLS001 that aims to meet the global corneal transplant demands.
Company: Cellusion Inc.
CEO: Shin Hatou, M.D., Ph.D.
Headquarters: 8-6 Nihonbashi-Kobuna, Chuo, Tokyo 103-0024, JAPAN
Founded: January 2015
URL: https://cellusion.jp/en/
For further information: please ask us through the contact form on our website.