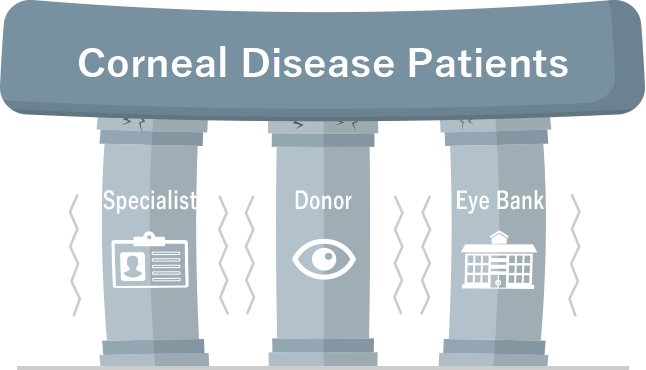
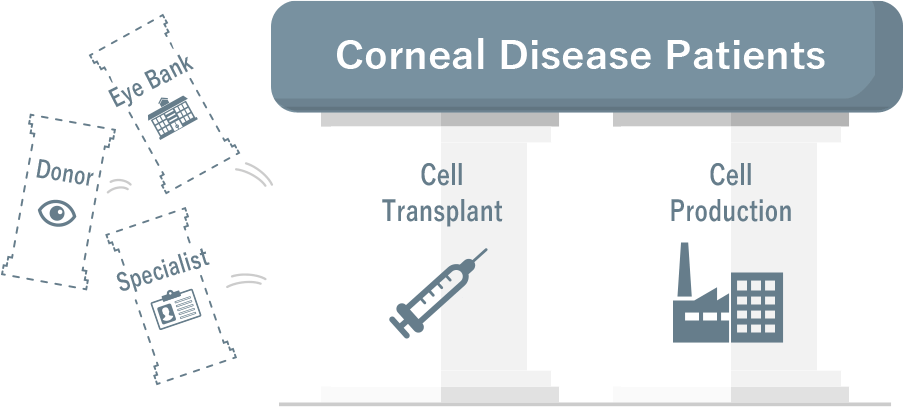
Cellusion Exhibited at JHVS 2024 and CEO Hatou participated in the “JHVS SHOWCASE”
2024. 10. 15
Cellusion exhibited at Japan Healthcare Venture Summit (JHVS) 2024 held at Pacifico Yokohama on October 9 to 11, 2024, and CEO Hatou took the stage at the “JHVS SHOWCASE” on October 10 to …